Lifechem Products
Silver trifluoromethanesulfonate ( Silver Triflate )
Trifluoromethanesulfonic Acid Silver Salt | Silver(I) Triflate | Silver Trifluoromethanesulphonate | Silver Trifluoromethanesulfonate | Ag(Otf),silver,trifluoromethyl)sulfonate | Agotf | Silver(I) Trifluoromethanesulfonate
| CAS No : | 2923-28-6 |
|---|---|
| EC Number : | 220-882-2 |
| Formula : | CAgF3O3S |
Product Specification
- Appearance & Physical State – light beige crystalline powder
- Molecular weight – 256.93700
- Boiling point – 162ºC at 760mmHg
- Melting Point – 356-357ºC
- Vapor Pressure – 1.14mmHg at 25°C
- Storage Condition – 2-8°C
- Stability – Stable, but may be light sensitive. Incompatible with strong acids, strong oxidizing agents.
Safety Information As Per GHS
Hazard Statement(s) as per GHS
- H315: Causes skin irritation.
- H318: Causes serious eye damage.
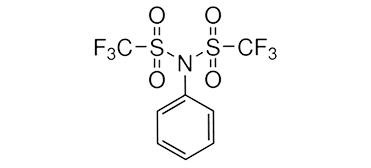
Silver Trifluoromethanesulfonate ( Silver Triflate )Silver trifluoromethanesulfonate, or silver triflate is the triflate (CF3SO3-) salt of Ag+. It is a colorless or
white solid that dissolves in water, as well as organic solvents, including benzene. The reagent is utilized in the
production of organic triflates, as well as inorganic triflates.Trifluoromethanesulfonate is a weakly coordinating anion, silver trifluoromethanesulfonate is useful as a halide
abstraction reagent.Reactive trifling agent, and source of silver ions that are soluble and useful to form electrophilic onium species
and for the stimulation of Friedel-Crafts, nucleophile alkene cyclization or esterification processes).Olefins are produced from secondary phosphates and Thiophosphates. With this reagent, the etherification process of
alcohols using primary alkyl halides can be done under mild conditions.Utilized to make the cationic rhodium catalysts derived from complexes of color rhodium to hydrophosphinate
Acetylenes. It can also be used as a catalyst in the preparation of silyl-ethers through hydrosilylation of
aldehydes.









Recent Comments