Lifechem Products
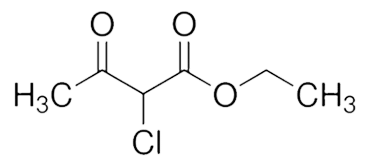
Ethyl-2-chloroacetoacetate
ethyl 2-chloro-3-oxobutanoate
Ethyl-2-ChloroacetoacetateEthyl chloroacetate is a chemical substance utilized mainly within the industry of chemicals. It is employed as a solvent in organic synthesis, as well as to act as an intermediate in the creation of pesticides (such as sodium fluoroacetate). A good example of the use of this chemical was in the production of Cinepazet.
Saccharomyces cerevisiae reduces beta-keto ester ethyl 2 chloroacetoacetate to equivalent chiral cis and trans-beta-hydroxy esters. As a result of chiral reductions, competing reduction of the substrate xenobiotic ethyl acetoacetate is triggered, in an action that is mediated by the cytosolic glutathione (GSH).
Mechanistically, dechlorination is a novel type of glutathione-dependent dehalogenation catalyzed by an as yet unidentified glutathione-dependent dehalogenase. The initial step is a nucleophilic substitution of the chloride substituent with glutathione.
In the following step that is catalyzed by an enzyme in the next enzyme-catalyzed step, a second glutathione mo releases the product of dehalogenation through an attack of thiolysis on the thioether bridge. This results in the net shift of electrons from the substrate, resulting resultant in the creation of glutathione disulfide (GSSG). It is effective in conditions of aerobic stress and catalyzed by the fungus, this deductive chlorination of an aliphatic substrate provides an excellent example of new, glutathione-mediated dehalogenation of microbial cells.
| CAS No : | 609-15-4 |
|---|---|
| EC Number : | 210-180-4 |
| Inchikey : | RDULEYWUGKOCMR-UHFFFAOYSA-N |
| Formula : | C6H9ClO3 |
Product Specification
- Appearance & Physical State – clear bright yellow liquid
- Molecular weight – 164.58700
- Boiling point – 200ºC
- Flash Point – 50ºC
- Smiles – C([C@@H](C(C)=O)Cl)(OCC)=O
- Density – 1.19
- Storage Condition – Keep away from heat, sparks, and flame. Keep away from sources of ignition. Store in a cool, dry, well-ventilated area away from incompatible substances.
- Water Solubility- 17 g/L (20 ºC)
Safety Information As Per GHS
- Hazard - 3.2 corrosive










Recent Comments